*This timeline primarily focuses on the AIDS epidemic as it was in the United States. For details on the origin of HIV/AIDS, click here.

1981
A cluster of patients in the United States are reported with a rare lung infection, Pneumocystis carinii pneumonia, and a rare type of cancer, Kaposi’s sarcoma. These conditions are usually linked to immunodeficiency disorders or old age. However, because these reports are made about young, previously healthy gay men, doctors assume that the immunodeficiency disorder is acquired.1
On July 3, 41 cases of Kaposi’s Sarcoma affecting gay men in New York and California are reported in the New York Times.1
By the end of 1981, there are 270 reported cases of severe immune deficiency in gay men, and 121 of these men have died.2
The U.S. Centers for Disease Control and Prevention (CDC) initially refers to the disease as GRID, gay-related immune deficiency disorder.
Government response is extremely limited during this period, however the federal government as well as the medical community saw a shift in prioritization from acute episodes of infectious disease to a focus on chronic, degenerative diseases, which would soon characterize HIV/AIDS.
1982-85
Researchers question if there is a direct link between the gay lifestyle and the disease. The media even nicknames the disease the “gay plague.” However, by 1982, there are cases of the disease in intravenous drug users, patients who received blood transfusions, and in children who contracted it from their mothers during pregnancy. The next year, AIDS is found in females whose male sexual partners had AIDS. These cases suggest that AIDS is transmitted either sexually or through exposure to blood or blood products (including contaminated needles and transfusions from HIV-contaminated blood donors).
In January of 1982, the first American AIDS clinic is established in San Francisco.1
On January 7th, the CDC reports cases in the female sexual partners of males with AIDS. This dispels the hypothesis that AIDS/HIV is solely a “gay” disease.1
On March 4th, the CDC notes that most cases of AIDS are reported among homosexual men with multiple sexual partners, Haitians, intravenous drug users, and hemophiliacs. This report suggests that the disease may be caused by an infectious agent that is transmitted via blood to blood contact and sexual intercourse.1
Later in the same year, the U.S. Centers for Disease Control and Prevention (CDC) establishes the National AIDS hotline to respond to public inquiries about the disease.1
On September 24, 1982, the CDC officially refers to the disease as Acquired Immune Deficiency Syndrome (AIDS) and defines it as, “a disease at least moderately predictive of a defect in cell-mediated immunity occurring in a person with no known case for diminished resistance to that disease.” The CDC later rules out transmission by casual contact as well as sharing food, water, air, or environmental surfaces.1
Additionally, in 1982, the first official case of AIDS in South Africa is reported, found in a South African man who contracted the virus while in California.
In 1983, the Pasteur Institute isolates the HIV virus for the first time.
AIDS clinics and support groups begin to form and in 1984, and cities like San Francisco, Los Angeles, and New York order bathhouses to close due to high-risk sexual activity. Blood banks begin screening the U.S. blood supply. The first known ‘safe sex’ publication, “How to Have Sex in an Epidemic,” is published by a gay American author and activist, Richard Berkowitz.2
Later in 1983, Congress passed the first bill that addressed the allocation of funds to the treatment and research of AIDS—$12 million for agencies within the U.S. Department of Health and Human Services (HHS).
During this year, AIDS epidemics are simultaneously developing in Europe in men who have visited the US as well as another incidence in people with ties to central Africa, with the beginning of investigations of occurrence of AIDS in African countries such as Rwanda, Zaire and others.
In September, the CDC identifies all major routes of HIV transmission (sexual contact, blood contamination, and mother to child transmission). This rules out the transmission by casual contact, food, water, air, or environmental surfaces.1
In 1985, the U.S. Food and Drug Administration (FDA) licenses the first commercial blood test, ELISA, to detect HIV in the blood. Blood banks also begin screening for HIV. To this day, ELISA still remains the most relevant blood test to detect HIV.1
By this time, at least case of AIDS has been reported from each region of the world.2
1986-89
The virus that causes AIDS is officially termed Human Immunodeficiency Virus (HIV).1
In 1987, the U.S. HIV Travel and Immigration Ban is passed. This ban disqualifies people with HIV from entering the United States.1
The U.S. Surgeon General releases a report encouraging parents and schools to talk about AIDS with their children and educate them on proper condom use. Later, he launches the first coordinated HIV/AIDS education campaign. For this campaign, he mails over 107 million copies of the booklet, Understanding AIDS, to American households.1
The first antiretroviral drug, zidovudine (AZT), is approved by the U.S. Food and Drug Administration (FDA). Later, dideoxyinosine (ddI) is introduced. The FDA allows for a swifter drug approval process in order to benefit persons with HIV/AIDS.1
Later in 1987, the FDA approves the Western blot blood test kit. This is a more specific test for HIV antibodies than ELISA. Both the western blot blood test and ELISA is still used today for detection of HIV antibodies in the blood.1
December 1, 1988 is marked as the first World AIDS Day. World AIDS Day is held each year with the goal of uniting people worldwide in the fight against HIV, showing support for those living with HIV, and commemorating those who have died. World AIDS Day was the first global health day.2
By 1989, the number of reported AIDS cases in the United States has reached 100,000.

1990s
In 1990, the U.S. Center for Disease Control and Prevention reports that during dental procedures, HIV transmission is possible. This is supported by a case in which a patient contracts HIV from an HIV-positive dentist during the dental procedure. Results from blood analysis, the HIV strand of the patient and the dentist was very similar. This leads to an increase in public debates on whether common dental and medical procedures with HIV-positive health professions is safe.1
Later in the same year, Congress enacts the Ryan White Comprehensive AIDS Resources Emergency (CARE) Act of 1990, which provides support via a federal grant of $220.5 million for community-based care and treatment services of HIV/AIDS, all within its first year. The program, which is the largest federal grant program for HIV in the country, is overseen by the Health Resources and Services Administration (HRSA). Additionally, the United States Congress introduces further acts providing humanitarian aid, one of which was the AIDS Housing Opportunity Act. This act creates the Housing Opportunities for People with AIDS (HOPWA) program the following year (1991), grants from which provide housing assistance to people living with AIDS.
In 1991, a red ribbon emerges as an international symbol of AIDS awareness.
In 1992, AIDS becomes the number one cause of death for men ages 25 to 44.1
The U.S. Food and Drug Administration approves the use of a 10-minute diagnostic test kit to be administered by health professionals to detect the present of HIV.1 This significantly reduces the time frame for diagnosing HIV and patients would be treated earlier. Also, this lowers the emotional stress level of patients anticipating their HIV results.
In June of 1993, Congress approves the NIH Revitalization Act, which gave the Office of AIDS Research primary oversight of all NIH (National Institutes of Health) AIDS research. This Act demands that the NIH as well as other research organizations expand their involvement of women and minorities in all areas of their study. In the same year, the FDA approves the female condom.2
By 1994, AIDS becomes the number one cause of death for all Americans ages 25 to 44. A world total of 1 million cases of AIDS, 18 million cases of HIV-positive adults, and 1.5 million HIV positive children are reported. AIDS is considered to be the fourth biggest cause of death worldwide and the number one cause of death in Africa.1
On January 1st of 1996, UNAIDS (the Joint United Nations Programme on HIV/AIDS) commences its operations. This committee is established to champion for international action to be taken on the epidemic, and additionally to establish cooperative efforts confronting HIV/AIDS in the United Nations system.
In 1996, the number of AIDS cases diagnosed in the U.S. declines for the first time since it was discovered. Aids are no longer considered the leading cause of death for all Americans age 25 to 44.1
In 1997, Highly Active Antiretroviral Therapy (HAART) becomes the standard of HIV care, and AIDS-related deaths in the U.S. drop by 47% compared to the previous year. However, by the end of the year drug resistance is becoming a common concern. 1 In the same year, the type of community-based and federally financed healthcare construction inaugurated by the Hill-Burton Act (1946) ends, although many Hill-Burton clinics continue to exist, providing care to low-income citizens.
The World Health Organization estimates that there are 33 million people living with HIV worldwide and 14 million people who have died of AIDS so far.1
2000s
In 2000, President Clinton signs an Executive Order to help underdeveloped countries low on treatments to import and produce the generic HIV treatments, making them more accessible to their citizens.
The average life expectancy in sub-Saharan Africa decreases from 62 years of age to 47 as a result of AIDS. President Clinton declares that HIV/AIDS is a threat to U.S. national security. He issues an order to assist developing countries in the import and production of HIV treatments. Several drug manufacturers agree to offer reduced drug prices to developing countries.2
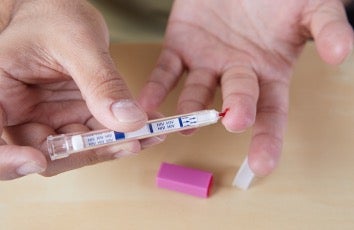
In November 2002, the FDA approves a rapid HIV diagnostic test kit that can be stored at room temperature. This test kit is 99.6% accurate and provides results in just 20 minutes, allowing HIV testing to become more widespread.1
In 2003 and 2004 (respectively), Congress allocates their first fund of $350 million to the United States President’s Emergency Program for AIDS Relief (PEPFAR) and UNAIDS commences The Global Coalition on Women and AIDS with a goal of increasing the visibility of the global impact the epidemic has had on women and girls.
A study at University of Illinois study finds that medical circumcision can reduce a male’s risk of acquiring HIV during heterosexual intercourse by 53%.2
In 2009, the Joint United Nations Program on HIV/AIDS reports that there has been a 17% decline in new HIV infections over the past decade. However, there has been a 25% increase in East Asia during this period. This increase is mainly due to its extensive size. The number of adults and children newly infected with HIV goes from approximately 74,000 in 2001 to 88,000 in 2010. In 2010, an estimated 0.1% of the East Asian population is living with HIV. 3
2010s
On January 4, 2010, the United States government officially lifts the HIV Travel and Immigration Ban. South Korea, China, Namibia, Armenia, Fiji, the Republic of Moldova, and Mongolia also lift their HIV travel restrictions.3
The HPTN 052 trial shows that early antiretroviral treatment reduces the risk of HIV transmission among couples by 96%.4
Fifty four percent of people eligible for antiretroviral treatment are currently receiving it.3
In 2012, the FDA approves the first HIV test that allows people to self-test at home.1 In the same year, the FDA also approves the use of Truvada as a means of pre-exposure prophylaxis (PREP). This is a medication for high risk people to take when they are HIV-negative in order to reduce their risk of future infection significantly.1

In 2013, Uganda passes the Anti-Homosexuality Bill, which enforces life imprisonment for both male and female homosexuals. There is a reported rise in violence and discrimination against homosexuals. Many individuals avoid HIV testing out of fear of being stigmatized.4
Later that year, on November 21, President Obama signs the HIV Organ Policy Equity Act (HOPE) which allows people living with HIV to receive organ donations from other infected donors. This HOPE Act provides the potential to save about 1,000 HIV-infected patients with liver and kidney failure annually.1
In 2014, the U.S. Food and Drug Administration approves the first diagnostic test that can differentiate between the two different HIV strains (HIV-1 and HIV-2). 1
In 2015, the CDC’s annual HIV Surveillance Report indicates that HIV diagnosis rates in the U.S. remained stable between 2009 and 2013.1
In 2018, an independent volunteer panel composed of experts in preventative medicine, The U.S. Preventive Services Task Force, sign a recommendation to clinicians to offer high-risk individuals PrEP. The panel gives its strongest recommendation to the drug, stating that it is highly effective and offers great promise to affected communities.
At a 2019 Conference on Retroviruses and Opportunistic Infections (CROI), investigators publicize the second observed case of an HIV-cured person, with no detectable infection three years after bone marrow transplant.1
Concluding Remarks
Since the first detection of cases in the United States in 1981, the HIV/AIDS epidemic took on many notable forms and required considerable resources to combat. Additionally, it rendered stigmatization to the gay male community, who were initially disproportionately affected by the disease. Through much clinical research, prevention mechanisms for HIV/AIDS as well as treatments for the conditions have been developed, including PrEP, PEP, and antiretroviral drugs. For more information on these options, one should consult a medical professional or facility. With proper knowledge and safety precautions, such as using a male condom, the spread of HIV/AIDS is preventable.
References
1. “A Timeline of AIDS.” AIDS.gov. US Department of Health and Human Services, 2012. Web. 02 Jan. 2015.
2. Mandal, Ananya, M.D. “History of AIDS.” News-Medical.net. AZO Network, 2 Feb. 2014. Web. 04 Jan. 2015.
3. Superville, Darlene. “HIV Travel Ban Lifted By President Obama.” The Huffington Post. TheHuffingtonPost.com, 24 July 2012. Web. 04 Jan. 2015.
4. UNAIDS World AIDS Day Report 2011. Geneva: UNAIDS, 2011. UNAIDS. 2011. Web. 5 Feb. 2015.
Last Updated 4 March, 2021.
