Pre-exposure prophylaxis (PrEP) is an HIV prevention method that involves HIV-negative individuals at risk of HIV infection taking a daily medication before being exposed to the virus. “Pre-exposure” means before coming into contact and “prophylaxis” stands for preventative treatment.1 At the time this article was updated, only two medications, under the brand names Truvada® and Descovy®, have been approved by the U.S. Food and Drug Administration (FDA) for daily use to prevent HIV infection.2 The U.S. FDA also approved several generic versions of Truvada® (emtricitabine 200 mg/tenofovir disoproxil fumarate 300 mg), which are considered equivalent to Truvada® and have the same clinical effects and safety profiles.7 In this article, “PrEP” refers to both brand name and generic versions of Truvada®, and Descovy® unless specified.

Table of Contents
How Does PrEP Work?
PrEP regimens, including both Truvada® (emtricitabine and tenofovir disoproxil fumarate) and Descovy® (emtricitabine and tenofovir alafenamide), are combinations of two different anti-viral drugs that lower the chance of HIV-1 infection by interfering and stopping the virus from duplicating and spreading in the host’s body.1 PrEP is not a vaccine and must be taken once each day consistently. A certain amount of PrEP must be present in the bloodstream for it to be effective. PrEP reaches maximum protection against HIV acquired through receptive anal sex when an individual has been taking the medication daily for about 7 days. Truvada® reaches maximum protection after about 21 days of daily use for receptive vaginal sex and for injection drug use.2 Descovy® has not been studied for HIV prevention in the case of receptive vaginal sex.
How Effective Is PrEP?
PrEP is a highly effective HIV prevention method when taken daily and consistently. According to current studies, PrEP reduces the risk of HIV infection through sex by approximately 99% when taken consistently.2 For people who inject drugs, PrEP reduces the risk of HIV infection by at least 74% when taken consistently.2 Inconsistent use or missing a dose decreases the effectiveness of PrEP. The risk of getting HIV from sex can be further lowered when PrEP is combined with other preventive methods, such as condom use.2 PrEP is one of the most effective HIV prevention methods when used correctly. It is the individual’s responsibility to follow their health professional’s instructions and to take PrEP as indicated.

Who Is PrEP Suitable for?
PrEP is suitable for people who do not have HIV, who weigh at least 35 kg (77 lbs), and who are at high risk of HIV exposure through sex or drug injections.1 Truvada® for PrEP is recommended for all people at risk of HIV through sex or through injecting drugs. Descovy® for PrEP is recommended for people at risk of HIV only through sex, excluding people who have receptive vaginal sex.
According to the Centers for Disease Control and Prevention’s (CDC) guidelines, people at high risk of HIV infection include, but are not limited to, the following:
Individuals who have had anal or vaginal sex within the past 6 months and:
- Have an HIV-positive sexual partner (especially if the partner has an unknown or detectable viral load).
or - Have not consistently used a condom.
or - Have contracted an STI within the past 6 months.
Individuals who inject drugs and:
- Have an HIV-positive injection partner.
or - Share needles, syringes, or other equipment to inject themselves.
Individuals who have been prescribed non-occupational post-exposure prophylaxis (PEP) and:
- Report continued risk behavior.
or - Have used multiple courses of PEP.
Individuals need to be tested for HIV before taking PrEP and at least every three months while taking PrEP. Individuals need to maintain HIV-negative status before and while taking PrEP since PrEP by itself is not a complete treatment for HIV.
Do not take Truvada® or Descovy® for PrEP if you are HIV-positive or if you do not know your HIV status.3 Do not take Descovy® for PrEP if you are a person with a vagina or are at risk of HIV from receptive vaginal sex.4 Do not take PrEP if you think you have been exposed to HIV. Post-exposure prophylaxis (PEP) is a treatment option for someone who may have recently been exposed to HIV. PrEP does not prevent other sexually transmitted infections (STIs). Consistent condom use is recommended when taking PrEP to further lower the risk of acquiring HIV and other STIs.
Is PrEP Safe?
There have been no significant negative health effects observed in people who are HIV-negative and have taken PrEP for a prolonged period of time. If an individual has a hepatitis B virus (HBV) infection, the HBV infection may worsen after they stop taking PrEP.2 Health care providers test individuals for HBV before and while taking PrEP.
| PrEP | Truvada3 | Descovy4 |
| Common side effects | Nausea Dizziness Headache Stomach pain Decreased weight | Nausea Fatigue Headache Stomach pain Diarrhea |
| Possible side effects | Kidney problems Lactic acid buildup in the blood Liver problems Bone problems | Kidney problems Lactic acid buildup in the blood Liver problems |
How Do I Start PrEP?
Talk to your health care provider to see if PrEP is right for you. PrEP can only be prescribed by health care providers to HIV-negative individuals who are suitable for PrEP. Individuals should follow up with their doctors at least once every three months to test for HIV, HBV, liver/kidney functions, and other tests as necessary.1 Take PrEP as instructed (usually daily, with or without food). If you miss a dose, take the next dose as soon as possible. Do not take two doses at once to compensate. Do not stop taking PrEP without consulting with a health care professional first.
The cost of PrEP varies. Generic versions of Truvada® are available and cost less than the brand name version. The generic version of Emtricitabine / Tenofovir (brand name Truvada®) is just as effective as the brand name version.
A common treatment plan for starting regular PrEP or daily PrEP involves taking one tablet every 24 hours. The time required to reach maximum protection from HIV infection varies based on what form of exposure the individual may be subject to. It takes about 7 days for PrEP to be fully protective if an individual is having receptive anal sex and about 21 days in the cases of receptive vaginal sex and injection drug use.
On-Demand PrEP/ “2-1-1” Method
On-demand PrEP, or the “2-1-1” method, is an alternative use of PrEP for cisgender men who are at risk of contracting HIV through anal sex. This method is recommended by several health departments and organizations in the United States, Canada, and Europe for men who have sex with men (MSM).5 The 211 method provides the same level of protection against HIV infection through anal exposure in cisgender men as the daily method.6 The 211 method can be used for single-day use or multi-day use. This on-demand PrEP method only applies to PrEP use with Truvada®. If you are using Descovy®, please consult with a healthcare provider.
Individuals who have vaginal sex are ineligible for the 211 method.5 The 211 method is not approved for protecting against HIV exposure through vaginal sex or drug use. It is not recommended for the following individuals to use the 211 method as their primary HIV prevention method due to insufficient evidence6:
- Trans femme or transgender women
- Trans masculine or transgender men who have anal sex
- Cis women who have anal sex
- Those who are using estradiol or testosterone (for oral contraceptive, hormone replacement therapy, etc.)
Daily PrEP is still a viable option for those who are ineligible for the 211 method.
Single Day Use
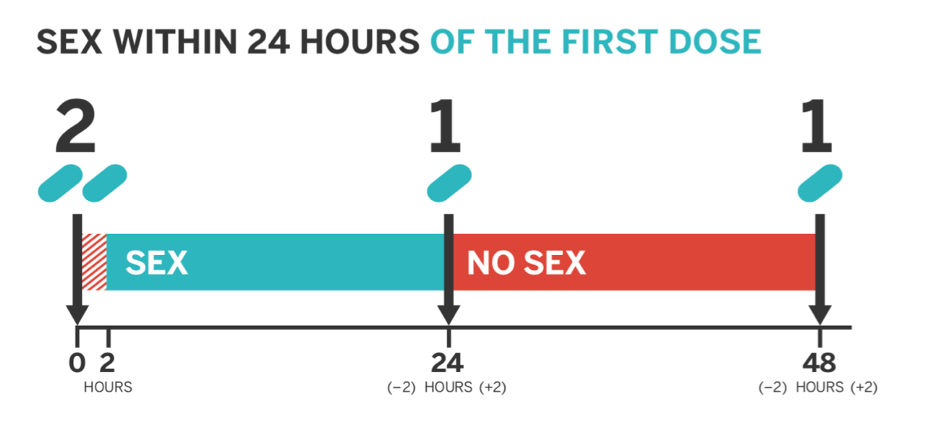
To initiate PrEP 211 dosing, individuals will take 2 emtricitabine/tenofovir(Truvada®) tablets 2-24 hours before anal sex, 1 tablet 24 hours after the initial dose, and finally an additional 1 tablet 48 hours after the initial dose.
Multiple Day Use
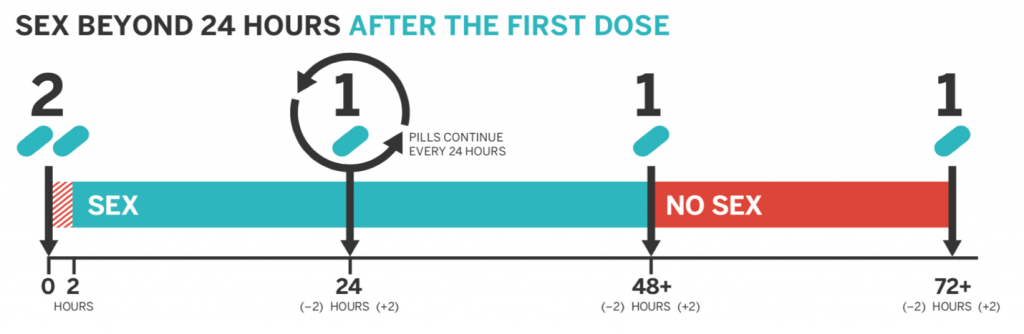
Multiple day use usually involves taking 2 emtricitabine/tenofovir(Truvada®) tablets 2-24 hours before anal sex and 1 tablet every 24 hours for as long as the individual is sexually active. To stop taking PrEP, individuals need to be sexually inactive for at least 48 hours and must not engage in sexual activity for the next 24 hours. Do not stop taking PrEP as long as you are sexually active or planning on having sex.
Concluding Remarks
Pre-exposure prophylaxis (PrEP) is a safe and highly effective HIV prevention method. In order to ensure maximum protection, PrEP must be taken daily on a regular basis or as instructed. Coupling PrEP with another prevention method, such as a condom, could reduce the chances of HIV infection even further. PrEP should only be taken by individuals who do not have HIV and have not been exposed to it. Depending on the person’s individual situation, a health care provider will recommend whether Truvada® or Descovy® can be used for PrEP. Consult with a health care professional before taking PrEP. Take PrEP as instructed and make sure to follow up with your doctor regularly. If you are experiencing side effects, or you want to stop taking PrEP, talk to your health care provider.
References
- “Information on HIV/AIDS Treatment, Prevention, and Research.” AIDSinfo, U.S. Department of Health and Human Services, 2020.
- “PrEP | HIV Basics.” Centers for Disease Control and Prevention, U.S. Department of Health and Human Services, 2019.
- TRUVADA for PrEP. Web. Gilead Sciences, 2020.
- DESCOVY for PrEP. Web. Gilead Sciences, 2020.
- “On-Demand PrEP.” Centers for Disease Control and Prevention, U.S. Department of Health and Human Services, 2021.
- “PrEP 211 Dosing Protocol.” San Francisco AIDS Foundation, 2019.
- “Cheaper Generic PrEP Now Available in the United States”. Web. POZ, 2021.
Last Updated: 25 May 2021.
